PDT is a quick, non-invasive, non-scarring, in-office treatment1-3
PDT with AMELUZ® (aminolevulinic acid HCI) topical gel, 10% and RhodoLED® red light can treat several AKs in one area2
Following PDT, most AK lesions will have cleared—revealing smoother-looking skin1

AMELUZ® is a topical gel used in combination with the RhodoLED® lamp series red light for PDT for the treatment of mild-to-moderate actinic keratosis on the face and scalp.2

RhodoLED® XL is a 5-panel LED lamp that emits a deep-penetrating red light that activates the PDT treatment.1,4,5


BF-RhodoLED® is an LED lamp that emits a deep-penetrating red light that activates the PDT treatment.1,4


AMELUZ® penetrates the epidermis of your skin to reach the premalignant cells that cause AK. It is then converted into the light-activated agent called PpIX. The red light of BF-RhodoLED® or RhodoLED® XL illuminates these cells and activates the agent, setting off a reaction that destroys premalignant cells that cause AK while leaving healthy skin cells mostly intact.1,2,6
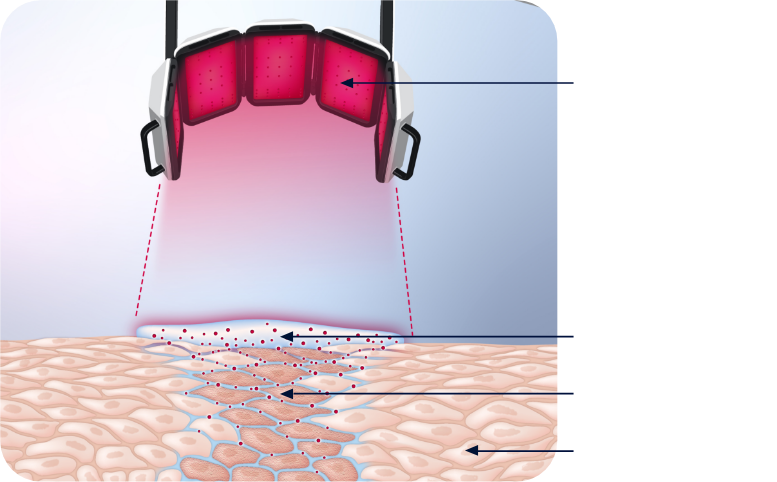
This is a visual representation of AMELUZ® PDT. Graphic is not designed to scale.
AMELUZ® penetrates the epidermis of your skin to reach the premalignant cells that cause AK. It is then converted into the light-activated agent called PpIX. The red light of BF-RhodoLED® or RhodoLED® XL illuminates these cells and activates the agent, setting off a reaction that destroys premalignant cells that cause AK while leaving healthy skin cells mostly intact.1,2,6
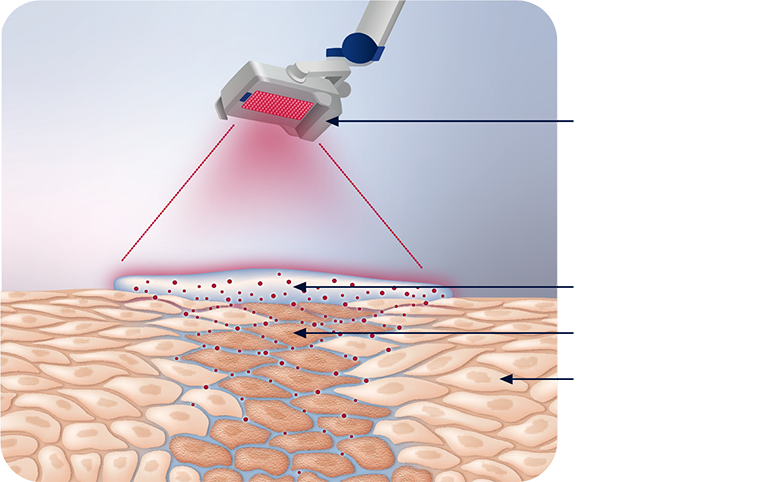
This is a visual representation of AMELUZ® PDT. Graphic is not designed to scale.
If you have any questions about AMELUZ®, please contact your health care provider.
If you have any questions about AMELUZ®, please contact your health care provider.
If you have any questions about AMELUZ®, please contact your health care provider.
INDICATION
AMELUZ® (aminolevulinic acid hydrochloride) topical gel, 10%, a porphyrin precursor, in combination with photodynamic therapy using BF-RhodoLED® or RhodoLED® XL lamp, is used for lesion-directed and field-directed treatment of actinic keratoses of mild-to-moderate severity on the face and scalp.
IMPORTANT SAFETY INFORMATION
Purpose: Photosensitizing agent
Uses: AMELUZ®, a porphyrin precursor, in combination with photodynamic therapy using BF-RhodoLED® or RhodoLED® XL lamp, is used for lesion-directed and field-directed treatment of actinic keratoses of mild-to-moderate severity on the face and scalp.
Warnings:
Do not use if you have a:
Ask your health care provider before use if you have:
When using this product:
Most common side effects at the application site are:
Most side effects occurred during illumination or shortly afterwards, were generally of mild or moderate intensity, and lasted for 1 to 4 days in most cases; in some cases they persisted for 1 to 2 weeks or even longer.
Pregnancy Warning: There is no available data on AMELUZ® use in pregnant women to inform a drug associated risk.
Lactation Warning: There is no available data regarding the presence of the active ingredient (aminolevulinic acid hydrochloride) in human milk, or the effects of aminolevulinic acid hydrochloride on the breastfed infant or on milk production.
Pediatric Warning: Safety and effectiveness in patients below the age of 18 has not been established.
Geriatric Warning: No overall differences in safety or effectiveness were observed between older (65 years and older) and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Directions:
Inactive ingredients: xanthan gum, soybean phosphatidylcholine, polysorbate 80, medium-chain triglycerides, isopropyl alcohol, dibasic sodium phosphate, monobasic sodium phosphate, sodium benzoate, and purified water.
References: 1. Reinhold U. A review of BF-200 ALA for the photodynamic treatment of mild-to-moderate actinic keratosis. Future Oncol. 2017;13(27):2413-2428. 2. AMELUZ [prescribing information]. Woburn, MA: Biofrontera Inc; 2024. 3. Benov, L. Photodynamic therapy: current status and future directions. Med Princ Pract. 2015;24(Suppl 1):14-28. 4. Agostinis P, Berg K, Cengel KA, et al. Photodynamic therapy of cancer: an update. CA Cancer J Clin. 2011;61(4):250-281. 5. Peng Q, Warloe T, Berg K, et al. 5-Aminolevulinic acid-based photodynamic therapy. Clinical research and future challenges. Cancer. 1997;79(12):2282-2308. 6. Maisch T, Santarelli F, Schreml S, et al. Fluorescence induction of protoporphyrin IX by a new 5-aminolevulinic acid nanoemulsion used for photodynamic therapy in a full-thickness ex vivo model. Exp Dermatol. 2010;19(8):e302-305.